Abstract
Background: Data that have been reported on antimicrobial use in Newfoundland and Labrador (NL) do not appear to be representative of use at the population level. We sought to use pharmacy network data on prescriptions to describe outpatient antimicrobial use in NL.
Methods: We analyzed all outpatient antimicrobial prescriptions dispensed between June 1, 2017, and June 8, 2021, from the provincial pharmacy network database and translated deidentified data into SPSS. We excluded prescriptions for parenteral and topical antimicrobials, antivirals and antifungals. We described antimicrobial use using the prescription rate and defined daily dose (DDD) rate.
Results: Overall, we analyzed 1 586 534 prescriptions dispensed to 394 708 people by 3431 prescribers. The rate of antimicrobial use was 741 prescriptions per 1000 population per year (7161 DDD/1000 population/yr). The median duration of prescriptions was 7 (interquartile range 7–10) days. The prescription rate decreased from 867 to 546 per 1000 population per year (−37%) over the study period, and the mean DDD rate decreased from 8387 to 5356 DDD per 1000 population per year (−36.1%). Antimicrobials with the highest DDD rate were amoxicillin (1568 DDD/1000/yr), doxycycline (864 DDD/1000/yr) and ciprofloxacin (633 DDD/1000/yr). Prescribers wrote a mean of 102 (standard deviation 248) prescriptions per year; 3 prescribers wrote more than 2500 prescriptions per year. Overall, 9203 (2.3%) of the 394 708 people in the study population received 4 or more prescriptions per year.
Interpretation: The rate of antimicrobial use in NL is lower than previously described in national surveillance data. Potential targets for stewardship intervention include prolonged duration of prescriptions, high-rate prescribers and high-rate patients, but further research is needed to assess the appropriateness of prescriptions according to diagnosis.
Antimicrobial resistance has been defined as one of the top 10 threats to global public health by the World Health Organization (WHO).1 Antimicrobial resistance was responsible for 5400 deaths and 880 000 days in hospital in Canada in 2018, costing the health care systems $1.4 billion.2 Although this problem is multifactorial, the main driver of antimicrobial resistance is antimicrobial use.3,4 Several provinces have reported data,5–7 but Canada does not currently have a national surveillance system for antimicrobial use.8 In 2020, the Canadian Antimicrobial Resistance Surveillance System (CARSS) report described Newfoundland & Labrador (NL) as having the highest rate of antimicrobial use in Canada.9 Further research also reports that people in NL use antimicrobials well above the national rate.10,11 This may be related to several factors such as the influence of nonmedical determinants of health or the high burden of chronic disease; NL has the highest provincial rates of obesity and smoking, as well as among the highest provincial rates of chronic kidney disease, chronic lung disease and diabetes. Compared with the national average of 41.7 years, NL also had the oldest population in Canada in 2022 at 45.3 years.12,13 Despite these factors, antimicrobial use in NL may not be as high as previously reported. Previous descriptions of antibiotic use in Canada have been done using databases from a third-party company — IQVIA, particularly the Canadian Compuscript Database and the Geographic Prescription Monitor Database — which uses sample data, not representative, population-based pharmacy network data.9,14
A comprehensive description of antimicrobial use is the starting point to inform interventions for antimicrobial stewardship in the outpatient setting, where most antimicrobial use occurs. We sought to analyze a population-level data set to comprehensively describe outpatient antimicrobial use in NL and identify potential areas for stewardship intervention.
Methods
Study design and setting
We conducted a retrospective, descriptive study of antimicrobial use, including all outpatient antimicrobial prescriptions written and dispensed to residents of NL between June 1, 2017, and June 8, 2021. Prescriptions from all physicians are included, regardless of specialty. Prescriptions from nurse practitioners and dentists were also included, as they were the only others with authority to prescribe oral antibiotics during the study period. We included only prescriptions that were dispensed. We also included prescriptions dispensed to patients in long-term care facilities.
Data source
We acquired prescription data from the Newfoundland and Labrador Centre for Health Information (NLCHI) Pharmacy Network database — a component of the provincial electronic health record and a province-wide database used by community pharmacy staff to record and share medication information. It contains patient-specific medication profiles, including demographic and drug information, prescribing physician and other related information for every NL community pharmacy and 3 hospital outpatient dispensaries. This encompasses all outpatient prescriptions dispensed in the province. Records in this database are internally validated by NLCHI, and data quality issues are addressed and monitored on an ongoing basis (https://www.nlchi.nl.ca/). This database has not been externally validated.
Records from the Pharmacy Network database were deidentified and health care numbers were consolidated using NLCHI’s internal deidentification process, then made available to researchers.
We extracted all prescription records for antimicrobial agents. We included only antibiotics identified by the 2022 WHO Collaborating Centre for Drug Statistics Methodology Anatomic Therapeutic Chemical (ATC)/Defined Daily Dose (DDD) Classification Index.15,16 We did not solely use antibiotics as our term of choice, we used the term antimicrobial use for ease of terminology. All antimicrobial agents included in the final analysis are listed in Appendix 1, available at www.cmajopen.ca/content/11/6/E1109/suppl/DC1. We excluded topical antimicrobials, antiviral agents, antifungal agents, antitubercular agents and parenteral antimicrobials.
Variables collected from each prescription included age, sex, postal code of patient’s home address, provider name and provincial drug coverage status. We changed 6-digit postal codes to 3-digit forward sortation areas for geographic analysis. We classified antimicrobials using the ATC/DDD Index.
We calculated the DDD using reference values from the ATC/DDD Index.15 Population denominators were based on Statistics Canada estimates for each year for both Canada and NL populations.13 We obtained populations of forward sortation areas from Statistics Canada’s 2021 Census.17 We defined urban forward sortation areas as all those encompassed within the St. John’s metropolitan area, and all others as rural.18
Data analysis
We used 2 measures of antimicrobial usage for analysis, namely the DDD and number of prescriptions. We calculated the rates of DDD and prescriptions per 1000 population per year for all included antibiotics, both individually and overall. We took special note of rates over time as they related to the COVID-19 pandemic. We evaluated the rates by antibiotic class (detailed in Appendix 1), WHO AWaRe (Access, Watch, Reserve) classification and forward sortation area. AWaRe is a tool developed by the WHO to support antimicrobial stewardship; these groups consider the impact of different antibiotics and antibiotic classes on antimicrobial resistance to emphasize the importance of their appropriate use.19 In addition, we calculated rates stratified by sex and age category. We standardized all outcomes by age and sex. We calculated the age- and sex-adjusted rates using the direct standardization method. These age adjustments used the Canadian population in 2021 as a referent and were based on the proportion of 5 age groupings (0–19 yr, 20–39 yr, 40–64 yr, 65–79 yr, ≥ 80 yr).
We grouped rates of antimicrobial use into 6 major classes (penicillins, cephalosporins, macrolides, tetracyclines, fluoroquinolones and other). We also grouped antibiotics into AWaRe classification groups.
We did not conduct any statistical testing. We used SPSS for data cleaning and analysis.
Ethics approval
Prescriptions were anonymized before analysis. The Health Research Ethics Authority of Newfoundland & Labrador determined that ethics approval was not required for this project.
Results
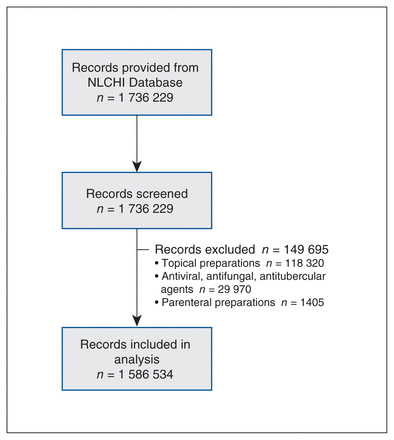
Overall, 1 736 229 dispensed prescriptions were in our database; we excluded 149 695 (8.6%), leaving 1 586 534 prescriptions included in our analysis (Figure 1). This represented 394 708 patients and 3431 prescribers. The mean age of patients was 47.7 (standard deviation [SD] 23.6) years. Sixty-one percent of prescriptions were dispensed to females. Twenty-six percent were beneficiaries of the provincial drug program, and 1.8% were admitted to long-term care. The mean duration of therapy was 10.4 (SD 11.9) days with a median duration of 7 (IQR 7–10) days (Table 1).
Study flow diagram. Note: NLCHI = Newfoundland and Labrador Centre for Health Information.
Patient demographic characteristics
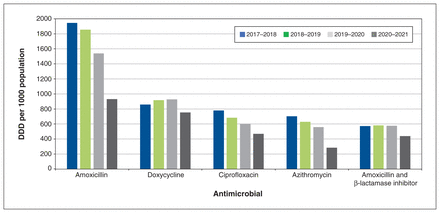
The mean DDD rate during the study period was 7161 DDD per 1000 population per year, which decreased by 36.1% over the study period. The first 3 years of the study period (before COVID-19) were responsible for only 14.1% of this decrease. The mean prescription rate over the study period was 741 prescriptions per 1000 population per year (Figure 2). The prescription rate decreased from 867 to 546 per 1000 population per year (−37.0%). The first 3 years were responsible for only 14.1% of this decrease. The 5 antimicrobials with the highest DDD rate were amoxicillin (1568 DDD/1000 population/yr), doxycycline (864 DDD/1000 population/yr), aiprofloxacin (633 DDD/1000 population/yr), azithromycin (544 DDD/1000 population/yr), and combination amoxicillin and β-lactamase inhibitor (543 DDD/1000 population/yr) (Figure 3). Usage of each individual antimicrobial declined overall. Usage of doxycycline and combination amoxicillin and β-lactamase inhibitor increased slightly in the first 3 years, then declined in the final year of the study period.
Antimicrobial prescriptions per 1000 population per year (age- and sex-standardized). Each point on the x-axis represents 1 calendar year (i.e., June–June).
Top 5 antimicrobials by age- and sex-standardized defined daily dose (DDD) per 1000 population per year.
Antimicrobial use by prescription rate was highest among people aged 80 years and older (1313 prescriptions/1000 population/yr). Antimicrobial use by prescription rate was lowest among those aged 20–39 years (522 prescriptions/1000/yr). The largest decrease in antimicrobial use was seen among those aged 0–19 years, both males and females (Figure 4). Females received more antimicrobial prescriptions than males in every age category. Antimicrobial use by prescription rate increased among patients aged 65–79 years and 80 years and older in 2019–2020 before declining again in 2020–2021.
Antimicrobial prescriptions per 1000 population per year for (A) males and (B) females by age category. *Each point on the x-axis represents 1 calendar year (i.e., June–June).
Ten percent of the study population received 2 or more antimicrobial prescriptions per year. More than 2% of the study population received 4 or more antimicrobial prescriptions per year, representing 14.8% of all prescriptions. Most prescriptions (69.1%) were from the WHO AwaRe Access category, with 30.9% from the Watch category and only 0.014% from the Reserve category.
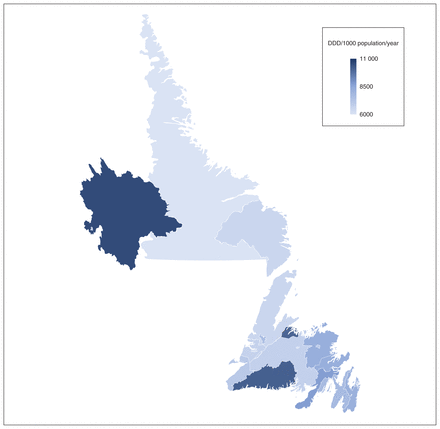
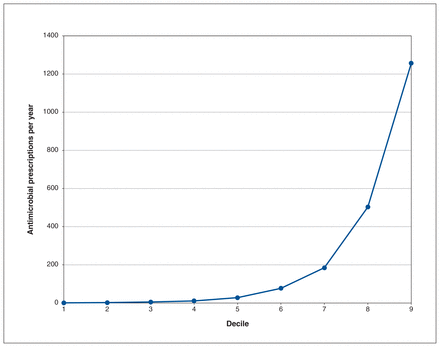
The range of rates among forward sortation areas was 5622–10 573 DDD per 1000 population per year. The top 3 areas were A0J, A0M and A0R, all of which were more than 2 SDs above the mean (Figure 5). The 3 lowest areas by DDD per 1000 population per year were A0P, A1C and A2H. The average among urban areas was 7251 (SD 779) DDD per 1000 population per year and the average among rural areas was 7814 (SD 1306) DDD per 1000 population per year. Prescribers in NL wrote an average of 102 antimicrobial prescriptions per year and 7 prescribers wrote more than 2000 antimicrobial prescriptions per year. The top 3 deciles wrote most antimicrobial prescriptions. The top 50 prescribers wrote more than 1000 antimicrobials prescriptions per year (Figure 6). Figure 7 describes antimicrobial use by class of antimicrobial.
Map of antimicrobial use (age- and sex-standardized defined daily dose [DDD] per 1000 population per year), stratified by forward sortation area.
Antimicrobial prescriptions per year by decile of prescribers.
Age- and sex-standardized defined daily dose (DDD) per 1000 population per year of antimicrobial prescriptions by class. *Each point on the x-axis represents 1 calendar year (i.e., June–June).
Interpretation
The findings of this population-based study of antimicrobial use in NL do not match with previous national reports. The rate for the 2019 period was reported by Crosby and colleagues10 as 950.2 prescriptions per 1000 population per year in NL, but we observed a range of 745 to 820 prescriptions per 1000 population per year in 2018–2019 and 2019–2020, a difference of roughly 17.7%. Our results indicate that the rate of antimicrobial use in NL is close to that of other provinces in eastern Canada, although still among the highest in the country. This difference may be attributed to our use of representative, population-based data.
As many jurisdictions lack comprehensive population-based data on antimicrobial use, databases from third-party companies such as IQVIA are commonly used for research purposes. Although their databases and methods are proprietary, some characteristics of these databases have been reported. The IQVIA database referenced by the CARSS report contains data from 6000 of 10 000 community pharmacies, with the rest of the data being extrapolated using proprietary geospatial extrapolation methods.9 A paper validating an IQVIA antibiotic database mentions an important limitation, with “larger errors noted in physicians practicing in rural locations … as they collect less prescription data from rural locations.”14 Given the potential inaccuracy in rural populations (which make up most of NL) and the results from our data, we purport that the widely used IQVIA databases may be inaccurate with regard to antimicrobial use in NL.
The rate of antimicrobial prescriptions decreased 37% over the study period and the mean DDD rate decreased by 36.1%. Much of this decline was associated with COVID-19; in the period before COVID-19, the prescription rate decreased by 14.1%. Additional NL data showing a 9% decrease in the number of antimicrobials written by family physicians from 2016 to 2017 support this trend.20 Other provinces have reported similar decreases, both before and after COVID-19.6,21
Decreases in antimicrobial use during COVID-19 may have been caused by reduced access or willingness to access care, measures to interrupt respiratory viral transmission or decreases in transmission of other respiratory viruses because of SARS-CoV-2.22–25 Decreases in antimicrobial use before COVID-19 may have been created by interventions in antimicrobial stewardship already occurring in the province. The provincial Antimicrobial Stewardship Committee and Quality of Care NL have intervened in the community with physician audit and feedback, public campaigns on antimicrobial use and clinical decision support tools.26–28 Other explanations include demographic changes such as changes in attitudes toward antimicrobial use among younger people.29 Data from Saatchi and colleagues6 support this explanation, reporting a decline in antimicrobial use in BC since 2000, as well as a decrease in a similar time frame between 2015 and 2018. However, data from Manitoba show usage increasing in that province between 2011 and 2016.7 During the pandemic, Kitano and colleagues30 reported a decrease in usage in Ontario and Knight and colleagues31 showed a decrease nationally, consistent with our findings. Provincial-level data describing duration of antibiotic prescriptions were not readily available for comparison; however, we believe the NL rate to be high and the variability wide. This may be associated with long-duration prescriptions.32
The top 5 most prescribed antimicrobials in our study are consistent with other sources.7,9,33,34 Ciprofloxacin was the third most used drug at 633 DDD per 1000 population per year. Despite previous evidence showing decreased usage of this antimicrobial in NL,35 this rate remains high. This is concerning given that, in 2016, an advisory from the United States Food and Drug Administration stated that the harms of quinolones usually outweigh the benefits in uncomplicated infections, in addition to the high prevalence of potentially inappropriate first-line use in Canada.36,37 Ciprofloxacin was moved to special authorization status in NL in 2019,38 requiring justification for use among patients included in the prescription drug program. Use of doxycycline and ciprofloxacin appear higher than in other regions.9,34
Antimicrobial use decreased across all age groups. The smallest decrease was seen among those aged 80 years and older. The largest decrease was seen in the 0–19 year age group. At the beginning of the study period, males aged 0–19 years received more prescriptions than males aged 40–64 years; however, at the end of the period, they received the lowest amount, in line with data in the female population. This is similar to the age cohort data reported in Manitoba.7 Usage was high among patients aged 65–79 years and those aged 80 years and older at 35.1% and 73.4% higher than the general population, respectively. Possible explanations include physical distancing for children during the pandemic, lower levels of misuse among older adults or physician hesitancy to change practices among populations with more health concerns.
We identified several areas with high rates of antimicrobial use and wide variability by forward sortation area. We found that rural areas had higher usage than urban areas by 7.21%. This is lower than data from Quality of Care NL, which showed that the number of antibiotic prescriptions per 1000 people was 17% higher in rural areas compared with urban areas.39 The 3 areas with the highest rates and 2 of the 3 areas with the lowest rate postal codes were rural. This suggest that rates are higher in rural areas, but that these areas also have a wide variability of prescribing practices. High regional variability in NL may be related to small population sizes, which are more susceptible to outliers skewing the results. Only 50 prescribers in NL prescribe more than 1000 antimicrobial prescriptions per year. Data from Quality of Care NL supporting this finding show that, in 2018, 20% of family physicians wrote 56% of all prescriptions for oral antimicrobials.20
Potential areas of intervention in NL include prescription duration, high-rate patients, high-rate prescribers, and high-rate areas. Reinforcing existing stewardship interventions such as peer comparison, while bolstering others like public education campaigns, would help optimize usage, maintaining the recent reduction to combat antimicrobial resistance.
Limitations
Our study is limited by the lack of data on diagnosis. Therefore, we cannot comment on appropriateness of prescription. Our data included prescriptions written by out-of-province prescribers and 2.39% of prescriptions were not associated with any prescriber, which may have skewed the mean prescriptions per prescriber slightly downward. Since information on prescriber setting and number of patients seen was not readily available, we cannot comment on factors that may explain the vast outliers observed. Upward or downward trends in antimicrobial use among older age groups may be influenced by variance in annual burden of respiratory disease, making it difficult to draw conclusions from differences across age groups. Around 2.65% of prescriptions were associated with either an invalid or out-of-province postal code. Our database relies on pharmacists entering the prescription information and, as such, is vulnerable to human error.
Conclusion
Our results indicated that the rate of antimicrobial use in NL was 27% lower than reported in previous studies. The COVID-19 pandemic was associated with a significant reduction in antimicrobial use across all age groups and both sexes, coinciding with the onset of public health restrictions. However, prepandemic rates were among the highest in the country. Further research targeting appropriateness of prescriptions and long-term care inhabitants is needed in NL.
Acknowledgements
The authors acknowledge Newfoundland and Labrador Centre for Health Information and Quality of Care for providing all health administrative data for this project.
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: All of the authors contributed to the conception and design of the work; data acquisition, analysis and interpretation; and drafting the manuscript. All of the authors revised it critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This study did not receive any funding.
Data sharing: Because patient identifiers are not included, data can be shared publicly on request to the corresponding author.
Disclaimer: All inferences, opinions and conclusions drawn in this research are those of the authors and do not reflect the opinions or policies of the data steward(s) from Newfoundland and Labrador Centre for Health Information.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/6/E1109/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors