Abstract
Background: Clinical guidelines for hypertension were updated with lower blood pressure targets following new studies in 2015; the real-world impact of these changes on antihypertensive drug use is unknown. We aimed to describe trends in antihypertensive drug utilization from 2004 to 2019 in British Columbia.
Methods: We conducted a longitudinal study to describe the annual prevalence and incidence rate of use of 5 antihypertensive drug classes (thiazides, angiotensin-converting enzyme [ACE] inhibitors, angiotensin II receptor blockers [ARBs], calcium channel blockers and β-blockers) among BC residents aged 30–75 years. We also conducted a cohort study to compare the risk of discontinuation and switch or add-on therapy between incident users of the above drug classes. We used linkable administrative health databases from BC. We performed a Fine–Gray competing risk analysis to estimate subhazard ratios.
Results: Among BC residents aged 30–75 years (population: 2 376 282 [2004] to 3 014 273 [2019]), the incidence rate of antihypertensive drug use decreased from 23.7 per 1000 person-years in 2004 to 18.3 per 1000 person-years in 2014, and subsequently increased to 22.6 per 1000 person-years in 2019. The incidence rate of thiazide use decreased from 8.9 per 1000 person-years in 2004 to 3.2 per 1000 person-years in 2019, and incidence rates for the other drug classes increased. Incident users receiving thiazide monotherapy had an increased risk of discontinuing any antihypertensive treatment compared with ACE inhibitor monotherapy (subhazard ratio 0.96, 95% confidence interval [CI] 0.95–0.97), ARB monotherapy (subhazard ratio 0.84, 95% CI 0.81–0.87) and thiazide combination with ACE inhibitor or ARB (subhazard ratio 0.86, 95% CI 0.84–0.88), and had the highest risk of switching or adding on.
Interpretation: First-line use of thiazides continued to decrease despite a marked increase in incident antihypertensive therapy following updated guidelines; incident users receiving ARB monotherapy were least likely to discontinue, and incident users receiving thiazide monotherapy were more likely to switch or add on than users of other initial monotherapy or combination. Further research is needed on the factors influencing treatment decisions to understand the differences in trends and patterns of antihypertensive drug use.
Hypertension is a leading risk factor for cardiovascular disease and death, estimated to be attributable for 54% of strokes, 47% of ischemic heart disease cases and 13.5% of deaths worldwide.1,2 In Canada, hypertension affects approximately 23% of adults and, in 2010, accounted for 10.2% of direct health care costs.3,4 Treatment of hypertension is complex, and clinical practice guidelines, including recommendations on blood pressure targets and the efficacy and safety of drug therapies, continue to be updated following assessment of the available evidence.5
Currently, 5 antihypertensive drug classes are recommended for the initial treatment of individuals with hypertension and without other compelling indications,6 namely, thiazide-type and thiazide-like diuretics (“thiazides”), angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), dihydropyridine calcium channel blockers (CCBs) and β-blockers.6–8 In a Cochrane review, low-dose thiazides were shown to significantly reduce mortality and cardiovascular events, including stroke and coronary heart disease in individuals with moderate-to-severe primary hypertension, and the evidence supporting them were graded as high quality.9 Angiotensin-converting enzyme inhibitors and CCBs were shown to be similarly effective, though the evidence for them was graded as low-to-moderate quality.9 Additionally, single-pill combinations of an ACE inhibitor or ARB with a thiazide or CCB have been recommended as an initial therapy since 2017 for their effectiveness in improving cardiovascular protection, blood pressure control and treatment adherence, and reducing adverse effects.10
Previously, trends in antihypertensive drug use between 1999 and 2014 were shown to be responsive to changes in clinical practice guidelines and the timing of clinical trials.11 However, trends and patterns of antihypertensive drug use have not been recently evaluated in the context of current guidelines and evidence. More recently, clinical guidelines for hypertension were updated following new studies on the effect of intensive blood pressure lowering on cardiovascular outcomes. In 2015, authors of 2 meta-analyses concluded that intensive blood pressure lowering significantly reduced the risk of major cardiovascular events (by 14%12 and by 20%13) and all-cause mortality (by 9%12 and 13%13), and that the benefits were consistent across patient subgroups with different baseline comorbidities and blood pressure.12,13 In the same year, the Systolic Blood Pressure Intervention Trial (SPRINT) research group concluded that intensive lowering of systolic blood pressure targets to less than 120 mm Hg, compared with less than 140 mm Hg, resulted in a 25% reduction in major cardiovascular events and all-cause mortality among high-risk individuals without diabetes.14 In 2016, the Hypertension Canada Guidelines, for selected high-risk individuals only, lowered the diagnostic threshold from 140/90 to 130/80 mm Hg and recommended a systolic blood pressure target under 120 mm Hg.15 In 2017, the American College of Cardiology and American Heart Association (ACC/AHA) guideline on high blood pressure in adults redefined the diagnostic blood pressure threshold for hypertension from 140/90 mm Hg to 130/80 mm Hg regardless of individuals’ cardiovascular risk, lowered blood pressure targets to under 130/80 mm Hg and recommended earlier initiation of pharmacotherapy for low-risk individuals at a threshold of 140/90 mm Hg,7 although careful consideration of the potential benefits and harms of intensive blood pressure treatment is encouraged.7,15
These changes were expected to affect the prevalence and incidence of hypertension (i.e., reclassification of previously nonhypertensive individuals) and the use of antihypertensive drugs (i.e., initiation of pharmacotherapy in treatment-naïve individuals or intensification of treatment to reach blood pressure targets).16,17 Moreover, the different drug classes present varying efficacy and tolerability profiles that may affect their use. The occurrence of adverse effects is one of several key factors that influence treatment adherence,18 and poorly controlled blood pressure is a common reason for switching antihypertensive medications.19 Therefore, we sought to examine real-world patterns of persistence in relation to the initial drug therapies.
In this 2-part study, we aimed to describe trends in anti-hypertensive drug utilization in British Columbia over a 16-year period from 2004 to 2019, and compare patterns of discontinuation and switch or add-on therapy in incident users of antihypertensive drugs.
Methods
This longitudinal study was conducted in the province of BC. Residents are eligible for public coverage for medically necessary services under the province’s Medical Services Plan (MSP). Residents enrolled in the MSP are also eligible for the BC PharmaCare program, which covers the costs of eligible prescription drugs, medical supplies and pharmacy services.
Data sources
We used anonymized, individual-level and linkable administrative health databases of the BC Ministry of Health (listed in Appendix 1, Table S1, available at www.cmajopen.ca/content/11/4/E662/suppl/DC1). We obtained information on drug dispensings from the PharmaNet database, which contains records of prescriptions filled at community pharmacies. Information on diagnoses and medically necessary fee-for-service physician services were obtained from the MSP Payment Information database. We used the Discharge Abstract Database to obtain information on diagnoses and procedures during inpatient hospitalizations and the National Ambulatory Care Reporting System for information on ambulatory care visits. Information on demographic characteristics, health plan enrolment and death were obtained via the MSP Registration and Premium Billings database.
The study was reported using the Reporting of Studies Conducted Using Observational Routinely-collected Health Data checklist.20
Trends in antihypertensive drug utilization
We identified a source population of BC residents aged 30 to 75 years who were enrolled in the provincial health plan between Jan. 1, 2004, and Dec. 31, 2019. We excluded beneficiaries of the First Nations Health Authority and federal programs for whom we had no access to drug data. Individuals younger than 30 years and older than 75 years were excluded as young- and late-onset hypertension are associated with secondary hypertension due to conditions such as renovascular disease and hyperaldosteronism.6,21
From the source population, we identified prevalent and incident users of the following antihypertensive drug classes: thiazides, ACE inhibitors, ARBs, CCBs and β-blockers. We defined “prevalent use” as a dispensing of at least 1 antihypertensive drug during the year (including incident users), and “incident use” as a dispensing of an antihypertensive drug with no record of any antihypertensive drug (listed in Appendix 1, Table S2) dispensed in the previous 5 years (Jan. 1, 1999, or later). Prevalence was expressed as a percentage, computed as the number of prevalent users divided by the total number of individuals in the source population during the calendar year. Incidence was expressed as a rate per 1000 person-years, computed as the number of incident users divided by the total person-years of health plan enrolment for the source population during the calendar year. Utilization trends of antihypertensives between 2004 and 2014 served as historical reference for trends observed following the meta-analyses of intensive blood pressure–lowering trials and SPRINT published in 2015, and the subsequent updates to clinical practice guidelines from Hypertension Canada (2016) and the ACC/AHA (2017).
Discontinuation, switch or add-on therapy
Incident user cohort
From the source population, we constructed an incident user cohort of individuals who initiated antihypertensive treatment on thiazide monotherapy, ACE inhibitor monotherapy, ARB monotherapy, combination thiazide with ACE inhibitor or ARB, or CCB between Jan. 1, 2004, and Dec. 31, 2014. Incident users were not required to have a record of a hypertension diagnosis to be eligible for inclusion. Cohort entry occurred 91 days after initial dispensing (explained below), and the cohort members were followed until Dec. 31, 2019. Individuals were excluded from the cohort if they were not continuously enrolled in the provincial health plan during the previous 2 years, had missing information on age or sex, were younger than 30 years or older than 75 years, died before cohort entry, received both an ACE inhibitor and ARB at initial dispensing, or were diagnosed with cancer (anticancer therapy is associated with the development of hypertension22), renal failure, secondary hypertension or other conditions indicated for the antihypertensive drug classes (Appendix 1, Table S3) at any time before cohort entry. Individuals were also excluded if they discontinued antihypertensive therapy between initial dispensing and cohort entry.
Endpoints
Two primary endpoints were assessed: discontinuation of any antihypertensive therapy, and switch to or add-on of a different antihypertensive drug class. Discontinuation was assigned using the refill-sequence model,23 in which the first medication-free gap of 90 days (1.5 times the median 60-day supply of antihypertensive drug prescription observed in BC) for any antihypertensive drug was considered discontinuation of antihypertensive therapy (Appendix 1, Table S2). Discontinuers were not followed beyond the first instance of a 90-day gap in therapy, and the discontinuation date was defined as the expected date of the next prescription refill. We did not adjust for stockpiling. Individuals who switched between antihypertensive drug classes before a 90-day gap were considered persistent users. Death before discontinuation was considered a competing event. Individuals who were event-free were censored at the end of health plan enrolment or end of follow-up (Dec. 31, 2019).
Switch or add-on therapy was defined as the first dispensing of an antihypertensive drug class different from the initial drug class (Appendix 1, Table S2). Switching from a combination product to its different components was not considered a switch or add-on. Discontinuation of any antihypertensive therapy or death before a switch or add-on were considered competing events. Individuals who were event-free were censored at the end of health plan enrolment or end of follow-up (Dec. 31, 2019).
Statistical analysis
We used Fine–Gray competing risk regression models to estimate the effect of initial drug class on the subdistribution hazard (“subhazard”) of discontinuation and the subhazard of a switch or add-on, accounting for competing events as defined above, where the null hypothesis was no difference between the initial drug class groups. The subhazard is the instantaneous rate of occurrence of an event in those who remain free of that particular event at a given interval.24 Individuals who switched or added on between initial dispensing and cohort entry were excluded from the switch or add-on analysis. Subhazard ratios were reported using thiazide monotherapy as the reference group. Multivariable models included covariate adjustment for age, sex, income level, provincial drug coverage and geographical area (defined in Appendix 1, Table S4). Sensitivity analysis was conducted using 60-day and 120-day gaps to define discontinuation. Competing risk regression analyses were performed using SAS Enterprise Guide version 7.15, and cumulative incidence functions were computed and plotted using the cmprsk and ggplot2 packages in R software version 3.6.1.
Ethics approval
The study protocol was approved by the University of British Columbia Clinical Research Ethics Board (H19-03491).
Results
Trends in antihypertensive drug utilization
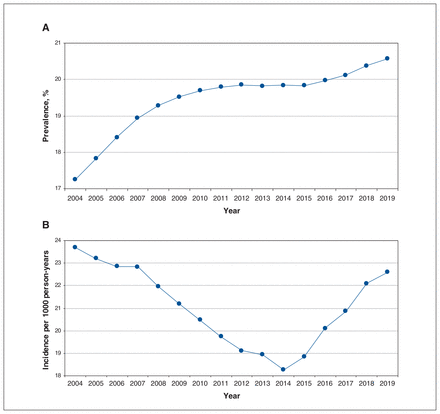
Among BC residents aged 30 to 75 years (range 2 376 282 individuals [in 2004] to 3 014 273 individuals [in 2019]), the overall prevalence of the 5 antihypertensive drug classes increased from 17.2% in 2004 to 20.6% in 2019 (Figure 1). Prevalent use of ACE inhibitors, the most prescribed drug class throughout the study period, increased from 8.7% in 2004 to 10.1% in 2019. Prevalent use of ARBs and CCBs also increased, for ARBs from 3.1% in 2004 to 5.7% in 2019, and for CCBs from 2.9% in 2004 to 6.0% in 2019. Prevalent use of thiazides increased in the first 5 years from 8.5% in 2004 to 9.5% in 2009, and subsequently decreased to 6.9% in 2019 (Figure 2).
Prevalence and incidence of antihypertensive drug use among residents of British Columbia aged 30–75 years between 2004 and 2019. Prevalence was computed as the number of prevalent users of thiazides, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, calcium channel blockers or β-blockers divided by the number of BC residents aged 30–75 years during the calendar year. Incidence was computed as the number of incident users of the 5 antihypertensive drug classes per 1000 person-years of health plan enrolment among BC residents aged 30–75 years during the calendar year. Prevalent use was defined as at least 1 of the 5 antihypertensive drug classes dispensed during the year. Incident use was defined as 1 of the 5 antihypertensive drug classes dispensed in the absence of a record for any antihypertensive drug dispensed in the 5 years prior (drug list available in Appendix 1, Supplementary Table S2 [www.cmajopen.ca/content/11/4/E662/suppl/DC1]).
Prevalence of antihypertensive drug use among residents of British Columbia aged 30–75 years between 2004 and 2019, by drug class. Prevalence was computed as the number of prevalent users of an antihypertensive drug class divided by the number of BC residents aged 30–75 years during the calendar year. Note: ACE = angiotensin-converting enzyme, ARB = angiotensin II receptor blocker, CCB = calcium channel blocker.
The overall incidence rate for the 5 drug classes decreased in the first 10 years from 23.7 per 1000 person-years in 2004 to 18.3 per 1000 person-years in 2014, and subsequently increased to 22.6 per 1000 person-years in 2019 (Figure 1). Incident use of thiazides decreased from 8.9 per 1000 person-years in 2004 to 3.2 per 1000 person-years in 2019 (Figure 3). After 2014, incidence rates increased for ACE inhibitors from 7.7 per 1000 person-years in 2014 to 9.7 per 1000 person-years in 2019, and for ARBs from 1.2 per 1000 person-years in 2014 to 1.7 per 1000 person-years in 2019. Incidence rates increased for CCBs from 1.2 per 1000 person-years in 2004 to 4.0 per 1000 person-years in 2019, and for β-blockers from 6.5 per 1000 person-years in 2004 to 7.1 per 1000 person-years in 2019. In an ad hoc analysis of incident users on thiazides, the percentage initiating hydrochlorothiazide decreased from 88.4% in 2014 to 74.6% in 2019 in favour of chlorthalidone (increased from 2.4% in 2014 to 14.4% in 2019) and indapamide (increased from 9.2% in 2014 to 10.9% in 2019) (Appendix 1, Figure S1).
Incidence of antihypertensive drug use among residents of British Columbia aged 30–75 years between 2004 and 2019, by drug class. Incidence was computed as the number of incident users per 1000 person-years of health plan enrolment among BC residents aged 30–75 years during the calendar year. Incident use was defined as 1 of the 5 antihypertensive drug classes dispensed in the absence of a record for any antihypertensive drug dispensed in the 5 years prior (drug list available in Appendix 1, Supplementary Table S2 [www.cmajopen.ca/content/11/4/E662/suppl/DC1]). Note: ACE = angiotensin-converting enzyme, ARB = angiotensin II receptor blocker, CCB = calcium channel blocker.
Discontinuation, switch or add-on therapy
After predefined inclusion and exclusion criteria were applied to the source population, the incident user cohort consisted of 232 781 individuals who initiated antihypertensive treatment between 2004 and 2014 (Appendix 1, Table S5). Most incident users initiated ACE inhibitor monotherapy (n = 100 670 [43.2%]) and thiazide monotherapy (n = 86 008 [36.9%]).
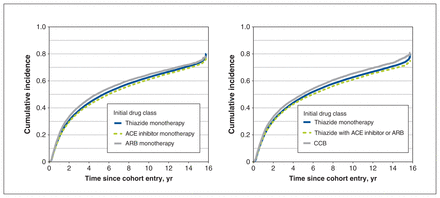
The median follow-up time for the discontinuation analysis was 5.0 (interquartile range [IQR] 1.4–9.4) years. The overall 1-year, 2-year, 5-year and 10-year cumulative incidence estimates of discontinuation were 0.18, 0.31, 0.48 and 0.62. Adjusted subhazards of discontinuation were reduced for individuals who started with thiazide with ACE inhibitor or ARB (subhazard ratio 0.86, 95% confidence interval [CI] 0.84–0.88) and individuals who started with ARB monotherapy (subhazard ratio 0.84, 95% CI 0.81–0.87) compared with thiazide monotherapy (Table 1, Figure 4). Adjusted analysis revealed that initiation with ARB monotherapy reduced the risk of discontinuation (subhazard ratio < 1) compared with initiation with thiazide monotherapy and ACE inhibitor monotherapy, whereas the unadjusted analysis showed the opposite (subhazard ratio > 1). Sensitivity analysis using alternate 60-day and 120-day definitions for discontinuation did not largely affect the magnitude or direction of these associations (Appendix 1, Tables S7 and S8).
Discontinuation of any antihypertensive therapy in the incident user cohort
Cumulative incidence of discontinuation of any antihypertensive therapy by initial drug class. Discontinuation was assigned using the refill-sequence model, in which the first medication-free gap of 90 days for any antihypertensive drug was considered discontinuation of antihypertensive therapy (drug list available in Appendix 1, Supplementary Table S2 [www.cmajopen.ca/content/11/4/E662/suppl/DC1]). The discontinuation date was defined as the expected date of the next prescription refill. The cumulative incidence estimates accounted for death as a competing event. Individuals who were event-free were censored at the end of health plan enrolment or end of follow-up (Dec. 31, 2019). Note: ACE = angiotensin-converting enzyme, ARB = angiotensin II receptor blocker, CCB = calcium channel blocker.
The incident user cohort for the switch or add-on analysis included 192 119 individuals (excluding 40 662 individuals who switched or added on before cohort entry), and the median follow-up time was 1.5 (IQR 0.5–4.5) years. The overall 1-year, 2-year, 5-year and 10-year cumulative incidence estimates of a switch or add-on were 0.24, 0.32, 0.45 and 0.53. Incident users on thiazide with ACE inhibitor or ARB had the lowest adjusted subhazard of switching or adding on (subhazard ratio 0.70, 95% CI 0.68–0.72), followed by CCB (subhazard ratio 0.78, 95% CI 0.76–0.80), ACE inhibitor monotherapy (subhazard ratio 0.84, 95% CI 0.83–0.85) and ARB monotherapy (subhazard ratio 0.94, 95% CI 0.90–0.97) (Table 2, Figure 5).
Switch to or add-on of a different antihypertensive drug class in the incident user cohort*
Cumulative incidence of a switch to or add-on of a different antihypertensive drug class by initial drug class. Switch or add-on therapy was defined as the first dispensing of an antihypertensive drug class different from the initial drug class (drug list available in Appendix 1, Supplementary Table S2 [www.cmajopen.ca/content/11/4/E662/suppl/DC1]). Switching from a combination product to its different components was not considered a switch or add-on event. The cumulative incidence estimates accounted for discontinuation and death as competing events. Individuals who were event-free were censored at the end of health plan enrolment or end of follow-up (Dec. 31, 2019). Note: ACE = angiotensin-converting enzyme, ARB = angiotensin II receptor blocker, CCB = calcium channel blocker.
Interpretation
Despite a marked increase in incident antihypertensive therapy from 2014 to 2019, initiation with thiazides continued to decrease while initiation with ACE inhibitors, ARBs, CCBs and β-blockers increased, potentially reflecting a shift away from thiazides as a preferred first-line therapy. Initiation with ARB monotherapy was associated with a lower risk of discontinuing antihypertensive therapy than initiation with thiazide monotherapy. Initiation with thiazide monotherapy presented the highest risk of switching to or adding on a different drug class, whereas initiation with a thiazide combination with ACE inhibitor or ARB presented the greatest reduced risk of switching or adding on compared with thiazide monotherapy. Although we are unable to determine reasons for discontinuation, switching or adding on in this study, we offer some possible explanations below.
Our finding of a decrease in incident antihypertensive therapy between 2004 and 2014 was consistent with a previously reported decrease in the age-standardized incidence rate of hypertension by 6 per 1000 population from 1999 to 2012 in BC.25 The increase in incident antihypertensive therapy from 2015 onwards corresponded in time with the publication of 2 meta-analyses of intensive blood pressure–lowering trials12,13 and SPRINT14 and the subsequent updates to Hypertension Canada (2016) and ACC/AHA (2017) guidelines that recommended lower blood pressure thresholds and targets.7,15 Muntner and coauthors estimated that implementation of the revised ACC/AHA blood pressure standards would increase initiation of pharmacological therapy by 1.9% and increase intensified treatment by 14.4% in existing users.17 We observed a 2.3% greater annual increase in incident antihypertensive therapy between 2017 and 2018 compared with the annual increase of the previous year. The increase in the prevalence of non-thiazide drug classes may be partially explained by the use of add-on therapy related to treatment intensification.
We observed a decrease in the prevalence and incidence of thiazide use consistent with previous studies.11,26,27 Contributing factors to the decrease may be related to concerns around potential adverse effects associated with thiazides (e.g., hypokalemia), 18,28 as well as evidence from the Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH; 2008) trial, which concluded that a combination of ACE inhibitor and CCB was superior in reducing adverse cardiovascular events in high-risk individuals compared with a combination of thiazide and ACE inhibitor.11,29 However, reasons for the continued decrease in thiazide use remains unclear. No new trials on first-line drugs for hypertension were identified in the 2017 update to the original Cochrane systematic review,9 and studies of real-world evidence on the comparative effectiveness and safety of antihypertensives provide further support for thiazides as a preferred first-line option.30,31 From 2017 to 2018, 3 Danish observational studies reported an association between hydrochlorothiazide and skin cancer.32–34 Following these studies, hydrochlorothiazide use decreased by 44% in Denmark.35 We did not observe a similar impact in BC; however, we found that the proportion of incident thiazide use with hydrochlorothiazide (thiazide-type diuretics) has decreased since 2014, as preference for chlorthalidone and indapamide (thiazide-like diuretics) has increased perhaps owing to their cardioprotective effects.6,36–38
Initiation with thiazide monotherapy was associated with a higher subhazard of treatment discontinuation than initiation with ACE inhibitor monotherapy, ARB monotherapy, and thiazide with ACE inhibitor or ARB, though the absolute differences were small. Previous observational studies showed similar directions of effect, where risk differences ranged from +8.9% to +32.8%, and they consistently reported a higher risk of discontinuation for thiazides than for other drug classes, and among the lowest risks for ARBs.30,39–42 Common reasons for nonadherence to antihypertensive treatment are the occurrence and progression of adverse effects, higher copayment and poor patient–provider relationship.18 In our study, incident users receiving ARB monotherapy had the highest subhazard of discontinuation before covariate adjustment, which was unexpected given good tolerability profiles of ARBs.43 Provincial drug coverage for ARBs differed from coverage for other drug classes; fewer than 9% of individuals receiving ARB monotherapy had coverage for their initial prescription, compared with 98% of individuals receiving other drug classes. Given that higher drug payment is associated with antihypertensive drug nonadherence18,44 and may be associated with choice of initial drug, drug coverage was included as a covariate for adjustment. After adjustment for all covariates, we observed a reversal of effect that showed superior persistence for ARB monotherapy, consistent with previous studies. Ad hoc analysis stratified by drug coverage showed the presence of Simpson’s paradox (Appendix 1, Table S6).45
Consistent with our study, 2 previous cohort studies found that initiation with thiazide monotherapy was associated with a higher risk of switching to or adding on a different antihypertensive drug class than initiation with an ACE inhibitor, ARB or CCB.30,39 These findings may suggest that less than optimal blood pressure control might be achieved with thiazide alone, thus leading to a switch to a different monotherapy or treatment intensification by the addition of 1 or more drugs to reach blood pressure targets.46 Additionally, we found that incident users receiving a thiazide combination with ACE inhibitor or ARB presented a significantly reduced risk of switching or adding on. Current guidelines recommend the use of an initial 2-drug (preferably single-pill) combination therapy for improved treatment efficacy, efficiency and tolerability compared with an initial monotherapy.6,7,47 A meta-analysis of 42 trials comparing the blood pressure–lowering effect of combination therapy versus monotherapy found that combining 2 drugs was 5 times more effective than doubling the dose of 1 drug.47 Initiation with combination therapy was also associated with a 34% risk reduction in cardiovascular events or deaths compared with initiation with monotherapy and a subsequent switch to combination therapy, owing to more rapid and effective blood pressure control. 48 Combination therapy may also provide more favourable tolerability profiles, as their individual components can be given at lower doses.46,47
Factors influencing treatment decisions are complex. It is unclear why thiazide use has declined given its effectiveness and potential for cost savings.9,49 Future research areas include evaluating the impact of prescriber and patient preferences on antihypertensive utilization, as well as real-world prescribing trends among individuals with specific clinical and demographic characteristics.50 Future studies on treatment persistence might also consider accounting for differences in drug coverage policies across drug exposures in their jurisdiction.
Limitations
A limitation of administrative data is the absence of clinical and laboratory data. Comorbid conditions, blood pressure measurements and biochemical markers are important considerations in individualized treatment decisions.51 Without these data, we could not evaluate utilization trends according to clinical characteristics, examine adherence to guidelines, ascertain reasons for discontinuation, switching or adding on, or confirm whether a switch or add-on was related to treatment intensification. For instance, common reasons for switching include insufficient blood pressure control, aiming for a better 24-hour effect and increase in cardiovascular risk.19 We did not follow individuals beyond the first occurrence of a discontinuation and switch or add-on and therefore did not examine whether discontinuers subsequently restarted treatment (either on the same or different drug class) or whether individuals experienced multiple switches or add-ons (which might indicate resistant hypertension). We did not adjust for seasonal variations in blood pressure, which may influence antihypertensive drug use and disease management.52 Additionally, individuals were not required to have a hypertension diagnosis; thus, patients with other conditions (e.g., β-blocker users with heart failure) may have been included, despite the exclusion criteria, and may have resulted in an overestimation of certain antihypertensive drug classes.
Conclusion
Incidence rates of antihypertensive therapy increased markedly after 2014, after the publication of new studies and updated guidelines recommending lower blood pressure thresholds and targets. Despite this, initiation with thiazides continued to decrease while initiation with other first-line antihypertensive drugs increased, potentially reflecting a shift away from thiazides as a preferred firstline therapy. Incident users receiving ARB monotherapy were least likely to discontinue antihypertensive treatment, and incident users receiving thiazide monotherapy were more likely to switch or add on than users of other initial monotherapy or combination. Further research is needed on the factors influencing treatment decisions to understand the differences in trends and patterns of antihypertensive drug use.
Acknowledgement
The authors thank Ellen Reynolds for editing a version of this manuscript. The BC Ministry of Health approved access to and use of BC data, facilitated by Population Data BC. All inferences, opinions and conclusions drawn in this manuscript are those of the authors and do not reflect the opinions or policies of the data stewards. The following data sources were used (https://www2.gov.bc.ca/gov/content/health/conducting-health-research-evaluation/data-access-health-data-central): BC Ministry of Health [creator] (2021): Medical Services Plan (MSP) Payment Information File. BC Ministry of Health [publisher]. BC Ministry of Health (2019); BC Ministry of Health [creator] (2021): PharmaNet. BC Ministry of Health [publisher]. Data Stewardship Committee (2019); Canadian Institute for Health Information [creator] (2021): National Ambulatory Care Reporting System. BC Ministry of Health [publisher]. BC Ministry of Health (2019); Canadian Institute for Health Information [creator] (2021): Discharge Abstract Database (Hospital Separations). BC Ministry of Health [publisher]. BC Ministry of Health (2019); BC Ministry of Health [creator] (2021): Consolidation File (MSP Registration & Premium Billing). BC Ministry of Health [publisher]. BC Ministry of Health (2019).
Footnotes
Competing interests: None declared.
This article has been peer reviewed.
Contributors: Anat Fisher and Colin Dormuth contributed to the conception and design of the study. Jason Kim and Anat Fisher contributed to the acquisition, analysis and interpretation of the data. Jason Kim drafted the manuscript, which all authors revised for important intellectual content. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: This project was funded by the British Columbia Ministry of Health, Pharmaceutical, Laboratory & Blood Services Division. All inferences, opinions and conclusions drawn in this manuscript are those of the authors, and do not reflect the opinions or policies of the British Columbia Ministry of Health.
Data sharing: The authors do not have permission to share data from this study. The data that support the findings of this study are available from Population Data BC (https://www.popdata.bc.ca) through the data access request process.
Supplemental information: For reviewer comments and the original submission of this manuscript, please see www.cmajopen.ca/content/11/4/E662/suppl/DC1.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/
References
- © 2023 CMA Impact Inc. or its licensors